|
11/14/2023 Has many complex proteins and so have a robust system for heat-shock protein assisted foldingRead Now 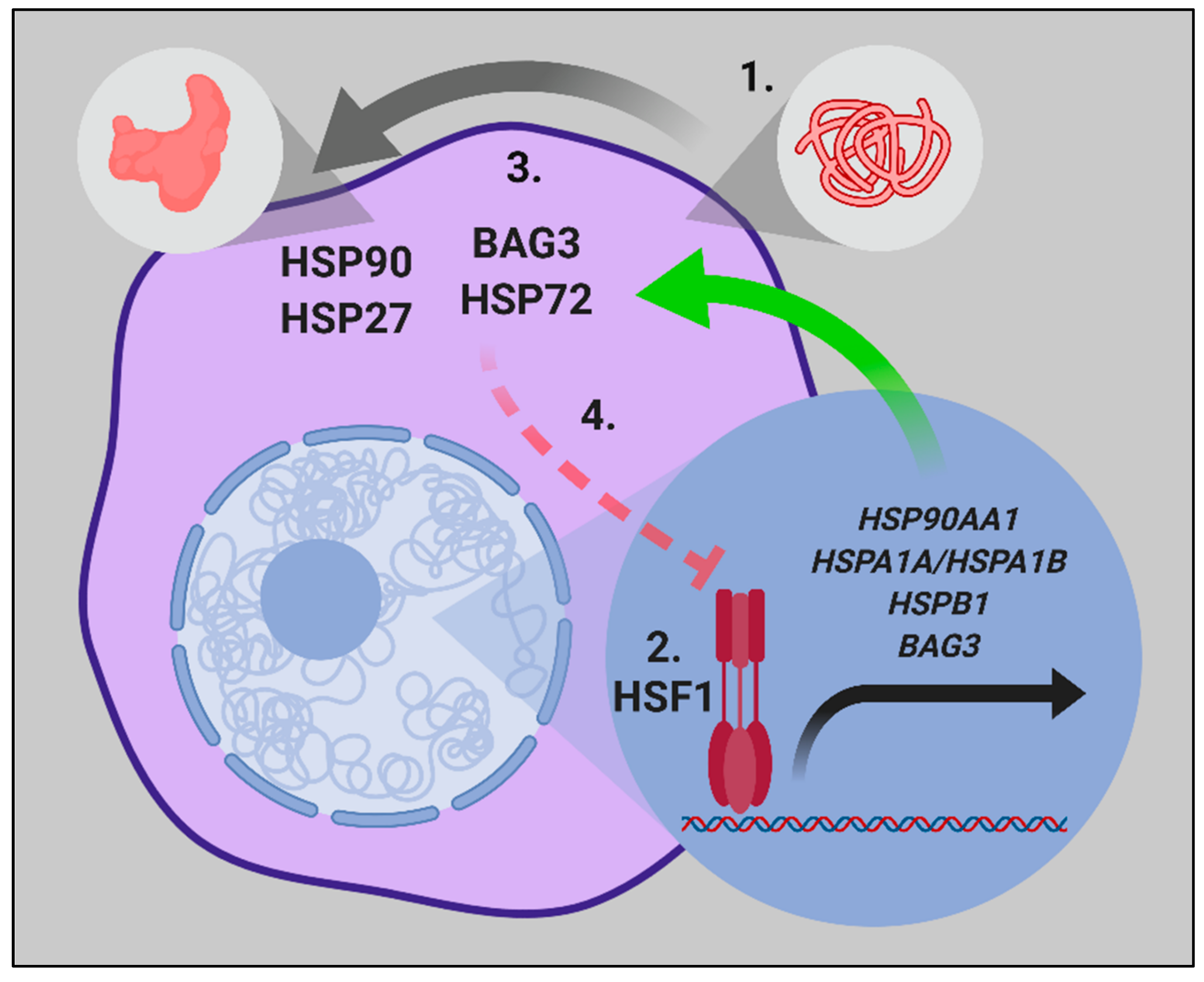
Georgopoulos (Eds.), The biology of the heat shock proteins and molecular chaperones (pp. Expression and function of the low-molecular weight heat shock proteins. HSPs antigen presentation chaperone heat-shock proteins.Īrrigo, A. This unit contains a table that lists common HSPs and summarizes their characteristics including (a) name, (b) subcellular localization, (c) known function, (d) chromosome assignment, (e) brief comments, and (f) references. HSPs constitute a large family of proteins that are often classified based on their molecular weight as Hsp10, Hsp40, Hsp60, Hsp70, Hsp90, etc. They also chaperone several toll-like receptors, which play a central role in innate immune responses. In addition, extracellular HSPs can stimulate and cause maturation of professional antigen-presenting cells of the immune system, such as macrophages and dendritic cells. More recently, HSPs have been shown to be key players in immune responses: during antigen presentation as well as cross-priming, they chaperone and transfer antigenic peptides to class I and class II molecules of the major histocompatibility complexes. Selected HSPs, also known as chaperones, play crucial roles in folding and unfolding of proteins, assembly of multiprotein complexes, transport and sorting of proteins into correct subcellular compartments, cell-cycle control and signaling, and protection of cells against stress and apoptosis. Heat-shock proteins (HSPs), or stress proteins, are abundant and highly conserved, present in all organisms and in all cells. 
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
